 Conclusions: Both the GRACE and TIMI risk scores were a good predictor of angiographic severity of CAD in patients with NSTE-ACS, and the GRACE score was found to be superior to the TIMI risk score.Ĭardiovascular diseases are currently the leading cause of mortality and morbidity in industrialized countries. Risk factors such as higher age, hypertension, smoking history, dyslipidemia, ECG changes such as ST deviation and T inversion, and Killip classification showed a statistically significant association with severity of disease. The area under the ROC curve for the GRACE score was 0.765 (95% confidence interval = 0.676-0.854), significantly superior to the area under the ROC curve of the TIMI score (0.715 95% CI = 0.618-0.812). Results: A positive association between the Gensini score and vessel score was observed with both the GRACE (P = 0.001) and TIMI (P = 0.001) scores. 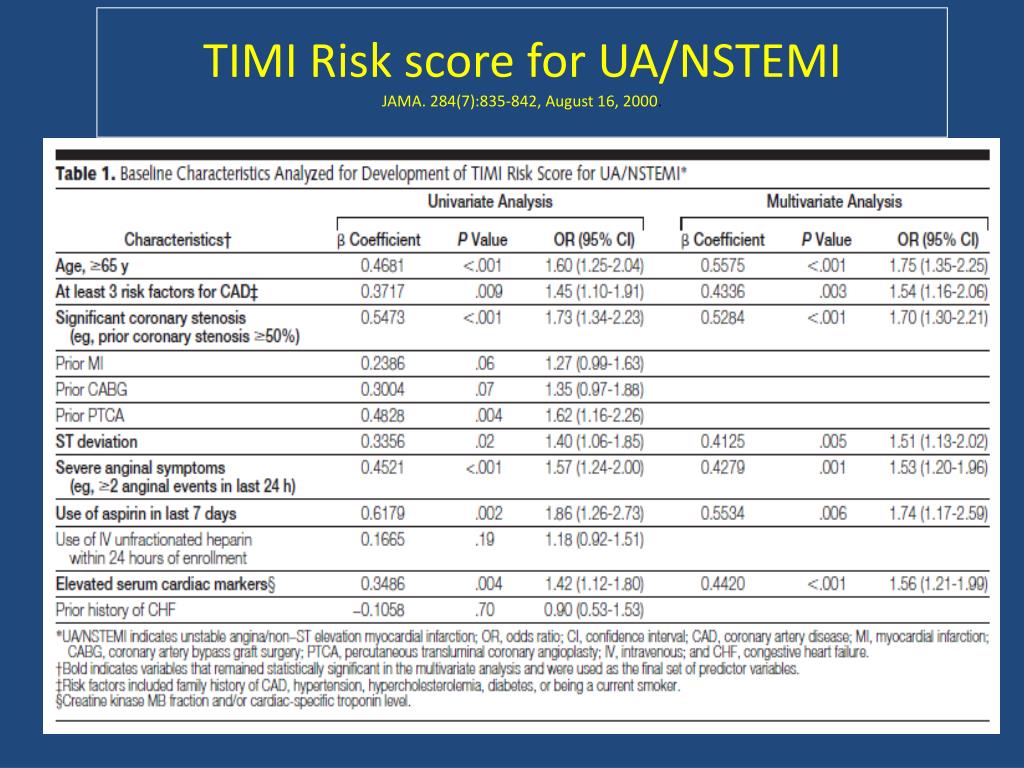
The receiver operating characteristic (ROC) curve was applied for the predictability of GRACE and TIMI scores for severity of disease. For comparison of two means, independent sample t-test/Mann-Whitney U-test was used, while for more than two means, one-way ANOVA/Kruskal-Wallis test was used. Statistical Analysis Used: SPSS software version 27.0 was used for statistical analysis. Coronary angiogram was done and the Gensini score and vessel score were used to assess the severity of CAD. The GRACE and TIMI scores were estimated. Subjects and Methods: Total 202 NSTE-ACS (NSTE myocardial infarction and unstable angina) patients were included. Settings and Design: This was an observational cross-sectional study. Aims: We aimed to compare the GRACE score with TIMI risk score for prediction of the angiographic severity of coronary artery disease (CAD) in patients with NSTE-ACS. SatapathyĬontext: The Global Registry of Acute Coronary Events (GRACE) and Thrombolysis in Myocardial Infarction (TIMI) scores in predicting coronary disease severity in patients with non-ST-elevation acute coronary syndromes (NSTE-ACS) have not been proved. 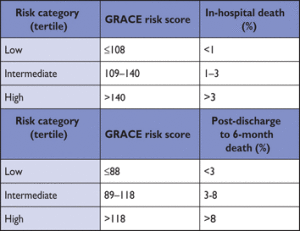
In patients with UA/NSTEMI, the TIMI risk score is a simple prognostication scheme that categorizes a patient's risk of death and ischemic events and provides a basis for therapeutic decision making. The slope of the increase in event rates with increasing numbers of risk factors was significantly lower in the enoxaparin groups in both TIMI 11B (P =.01) and ESSENCE (P =.03) and there was a significant interaction between TIMI risk score and treatment (P =. The pattern of increasing event rates with increasing TIMI risk score was confirmed in all 3 validation groups (P<.001). Event rates increased significantly as the TIMI risk score increased in the test cohort in TIMI 11B: 4.7% for a score of 0/1 8.3% for 2 13. The 7 TIMI risk score predictor variables were age 65 years or older, at least 3 risk factors for coronary artery disease, prior coronary stenosis of 50% or more, ST-segment deviation on electrocardiogram at presentation, at least 2 anginal events in prior 24 hours, use of aspirin in prior 7 days, and elevated serum cardiac markers. Outcomes were TIMI risk score for developing at least 1 component of the primary end point (all-cause mortality, new or recurrent MI, or severe recurrent ischemia requiring urgent revascularization) through 14 days after randomization. 
Relative differences in response to therapeutic interventions were determined by comparing the slopes of the rates of events with increasing score in treatment groups and by testing for an interaction between risk score and treatment. The TIMI risk score was derived in the test cohort by selection of independent prognostic variables using multivariate logistic regression, assignment of value of 1 when a factor was present and 0 when it was absent, and summing the number of factors present to categorize patients into risk strata. The 3 validation cohorts were the unfractionated heparin group from ESSENCE and both enoxaparin groups. A total of 1957 patients with UA/NSTEMI were assigned to receive unfractionated heparin (test cohort) and 1953 to receive enoxaparin in TIMI 11B 15 were assigned respectively in ESSENCE. Two phase 3, international, randomized, double-blind trials (the Thrombolysis in Myocardial Infarction 11B trial and the Efficacy and Safety of Subcutaneous Enoxaparin in Unstable Angina and Non-Q-Wave MI trial ). To develop a simple risk score that has broad applicability, is easily calculated at patient presentation, does not require a computer, and identifies patients with different responses to treatments for UA/NSTEMI. Patients with unstable angina/non-ST-segment elevation myocardial infarction (MI) (UA/NSTEMI) present with a wide spectrum of risk for death and cardiac ischemic events. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
