 Emission spectroscopy is a spectroscopic technique which examines the wavelengths of photons emitted by atoms or molecules during their transition from an excited state to a lower energy state. Schematic diagram of spontaneous emission. What is an emission spectrum and how is it formed?Īn emission spectrum is formed when an excited gas is viewed directly through a spectroscope. One example is astronomical spectroscopy: identifying the composition of stars by analysing the received light. The emission spectrum can be used to determine the composition of a material, since it is different for each element of the periodic table. What does an emission spectrum allow one to do? Liquid-sampling sources include flames and sparks (atom source), inductively-coupled plasma (atom and ion source), graphite furnace (atom source),… With the exception of flames and graphite furnaces, which are most commonly used for atomic absorption spectroscopy, most sources are used for atomic emission spectroscopy. What emission spectrum tells us? What is the source of atomic emission spectra? The number of neutrons and number of electrons are frequently equal to the number of protons, but can vary depending on the atom in question. 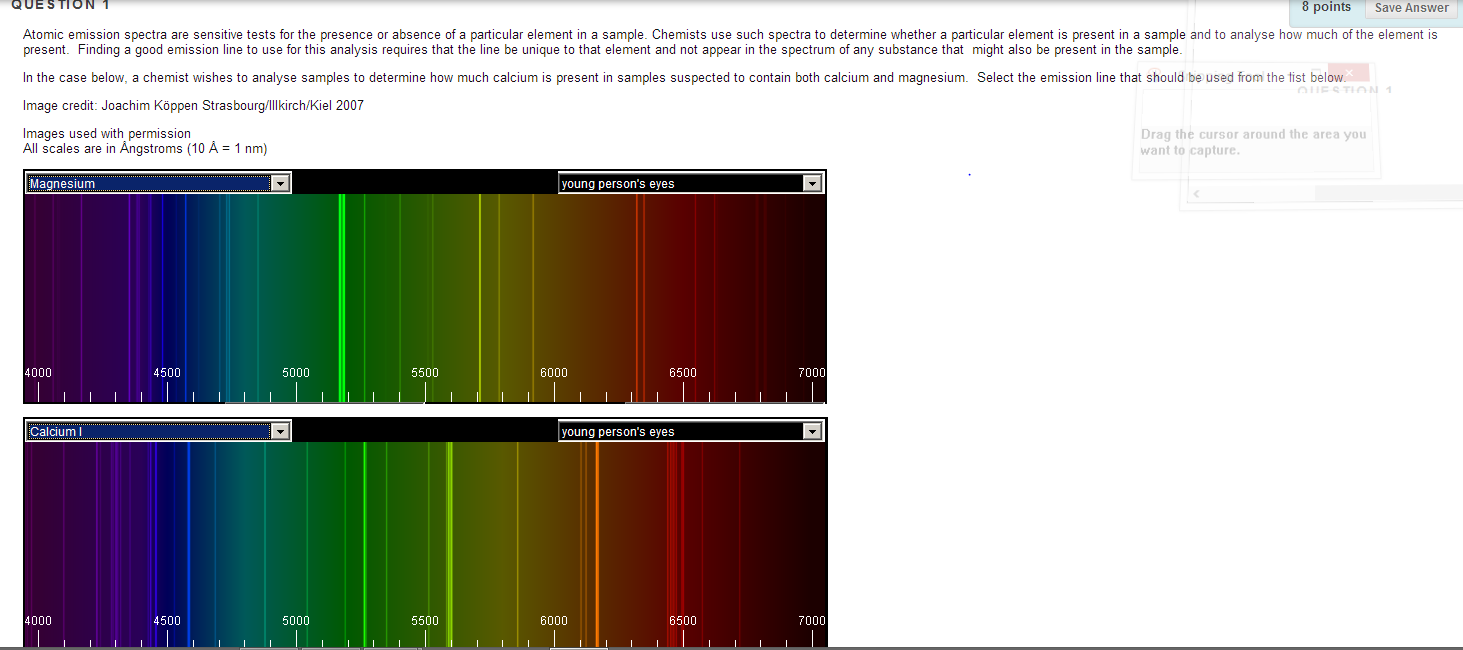
How do you determine the emission spectra element?Įxplanation: There are two properties that can be used to identify an element: the atomic number or the number of protons in an atom. The varying series of absorption and emission lines represent different ranges of wavelengths on the continuous spectrum. The emission spectrum of a chemical element or chemical compound is the spectrum of frequencies of electromagnetic radiation emitted due to an atom or molecule making a transition from a high energy state to a lower energy state.Īn emission line is formed when the electron falls back to a lower energy state, releasing a photon. Which of these is the best definition for an emission spectrum? By looking at the pattern of lines, scientists can figure out the energy levels of the elements in the sample. In emission spectra, bright lines will show up corresponding to the difference between energy levels of the elements, where in an absorption spectrum, the lines will be dark. How can you use the emission spectrum of an element to identify it? 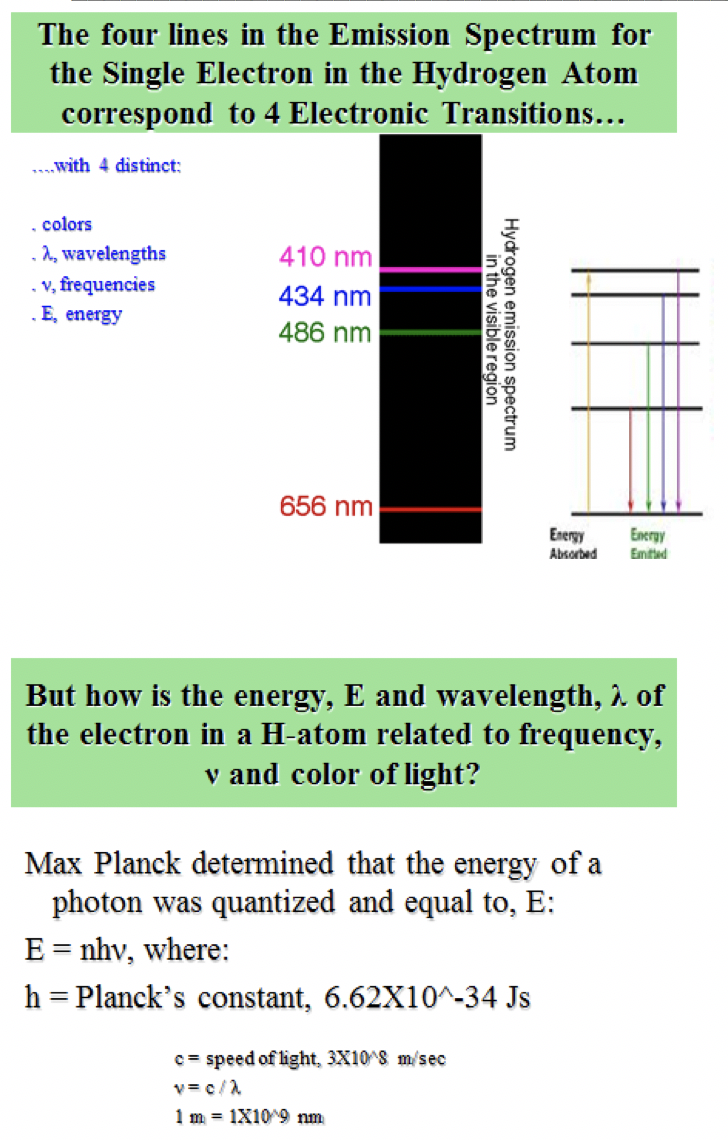
The spectral lines of a specific element or molecule at rest in a laboratory always occur at the same wavelengths. What creates the emission lines in a spectra for an element?Įmission lines occur when the electrons of an excited atom, element or molecule move between energy levels, returning towards the ground state. – The higher energy forms have shorter wavelengths and higher frequencies. – All forms travel at the same speed of light but have different wavelengths. Emission spectra are produced when photons are emitted from atoms as excited electrons return to a lower energy level. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
